
The safety and effectiveness of pharmaceutical products are the main responsibilities of biopharmaceutical organizations and global health authorities. A strict regulatory review and approval procedures are required to guarantee product safety, while the lengthy time frames needed for worldwide approvals cause delays in patients' access to life-saving medications. (Beierle et al., 2023). Integrating the electronic Common Technical Document (eCTD) with a Labeling Management System (LMS) is an important component of regulatory strategy. This paper provides a roadmap for integrating eCTD and LMS, transforming regulatory challenges into major strengths. As biopharmaceutical companies become more concerned about patients, sponsors and regulators thus prioritize patient safety. Many industries have already used modern technologies to improve speed and production. The biopharmaceutical business faces challenges in effectively utilizing existing technologies in a highly regulated environment. Regulatory success in the biopharmaceutical sector depends on the exchange of electronic or physical records, which limits communication with health authorities (Beierle et al., 2023).
The electronic Common Technical Document, or eCTD, is a format of document for sending regulatory information to the health authorities, including reports, applications, and supplements. Moreover, it contains an XML supporting format that provides information about the submission and documents. The purpose of eCTD is to lessen the workload for the health authorities. It is a common format used by all regulatory bodies, it streamlines the submission process. The eCTD facilitates the communication of regulatory information between industry and agencies, as well as the production, review, lifecycle management.
The enormous volume of information from the complete submission is tracked by eCTD, including information about manufacturers, IDs, sending and receiving organizations (Patil & Jogalekar, 2023).
Labeling is one of the important official documents throughout the product lifecycle as it provides product information in terms of safety, efficacy, and quality.
There are two types of labeling:
Healthcare professional: Healthcare professional HCP labeling, as required by local regulations, is a medical summary of product information intended for use by HCPs. It typically includes more thorough information to describe the benefit-risk profile.
Patient labels: The Patient Labeling/Information Leaflet (PIL) is prepared in patient-friendly language (Matsui et al., 2023).
A Labeling Management System (LMS) is a software solution that is designed to comply with product labels, packaging, and inserts in global markets by developing, managing, and implementing them. The LMS uses:
Structured Content Management (SCM) to manage, and reuse structured, regulatory approved content, ensuring accuracy and consistency.
SCM has been previously applied to support other functional areas within the pharmaceutical industry, such as clinical data management, supply chain management, and labeling (Beierle et al., 2023).
The regulatory framework allows for electronic submission of information in eCTD format, as compare to the traditional paper-based submission. The transition to an electronic system has provided various benefits. In the future, electronic filings can attain standardization across data systems, as it is shown in the figure 1 (Beierle et al., 2023).
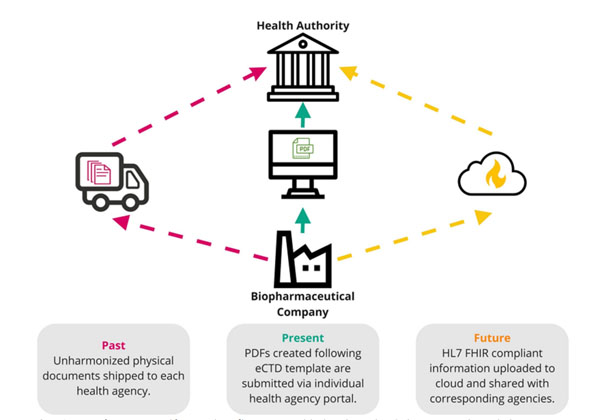
Figure 1. Summary of regulatory filing processes in past, present, and future
Labeling information is kept in a core dossier and then changed to meet region-specific regulations. These documents are living records that can alter based on factors such as the occurrence of a new adverse event or the approval of a novel use or dose. With that in mind, connecting eCTD with LMS could be a great achievement for the pharmaceutical industry.
Increased workflow efficiency: Avoiding repetitious use of content through multiple cycles of retyping and reorganizing data.
Reduced transcription errors: Health authorities analyze industry applicants' submissions and provide feedback in the form of data tables and text-based assessments written by the reviewer, thus increasing the chances of transcription errors.
Global regulatory requirements: The integration of eCTD (electronic Common Technical Document) with a Labeling Management System (LMS) promotes global regulatory compliance by making the medicine approval and labeling process faster, and more accurate worldwide.
Data integrity and version control: To maintain consistency among documents and data sets, both the preparation and assessment of regulatory submissions require rigorous and time-consuming processes to preserve data integrity and version control across documents.
Minimized laborious task: For instance, to view a regulatory submission for a legacy product while creating a new submission, a company must manually search for and interpret older documents to establish a frame of reference.
Increasing Automation and Opportunities for Machine Learning: Integrating eCTD with LMS solutions in biotechnology and pharmaceutical industries automates processes, enabling the use of machine learning techniques (Algorri, Cauchon, & Abernathy, 2020).
The following figure shows comparison that without Structured Content and Data Management (SCDM), regulatory Chemistry, Manufacturing, and Controls (CMC) submissions involve manual transcription, and multiple regional document variants. With SCDM, data is structured into content blocks, enabling flexible options like electronic submission and cloud-based transfer (Beierle et al., 2023).
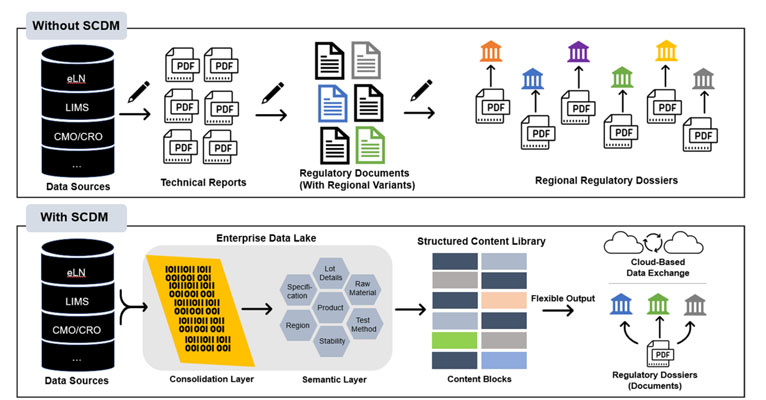
Figure 2. Comparison of with and without SCDM regulatory submission processes
Pharma leaders can minimize submission errors, save time, and ensure that patients and healthcare professionals receive the correct product information by using eCTD and LMS together. The following guide simplifies the process and provides recommendations to assist leaders successfully integrating eCTD with LMS (Macdonald, Isom, Evans, & Page, 2021).
Evaluate your current eCTD and LMS setups. Every system has its own set of specifications, which sometimes do not comply with the integration process and require some modifications. Consider the following before integration:
A specific format (e.g., XML, SPL) is needed to export data for LMS.
The latest regulatory requirements work with up-to-date eCTD submission tools.
Thoroughly identify gaps and determine whether you need a new system or if existing tools can be modified.
Set your goals or outcomes you want to achieve from integration, such as:
Submission errors that result from outdated labels can be reduced.
Real-time synchronization between LMS and eCTD submissions must be ensured.
The latest approved labels are always used in submissions by automating updates.
Stay up-to-date regarding guidelines of FDA, EMA, and ICH.
Manufacturers want label databases that can accommodate various label variants and can be updated in a timely and cost-effective manner. Secure platforms that allow authorities to exchange information between regulators and give transparency, and efficiency in approving products across multiple geographies. (Macdonald et al., 2021)
Automating regulatory operations can reduce the number of manual tasks required.
Automating workflows with the help of integration results in
Stay notified for missing or outdated documents.
The eCTD submissions will be automatically updated whenever a label change is approved.
Keep proper audit records.
For a smooth integration, the right digital tools make a great difference. Before integrating an LMS and eCTD software, consider the following factors:
Use of outdated data can be prevented by real-time version.
Support for XML-based submissions.
Any label changes and updates can be tracked automatically.
Use of Tools or systems that can ensure compliance with industry standards and global health authority requirements.
Proper training of the team is required for successful system. Regular training and will help the workforce in:
Integrated system can be used effectively after thorough understanding.
Know how to troubleshoot common issues.
Having updated knowledge on regulatory changes and software updates.
Provide funds for training in modern digital tools and technology. Replacing manual labor by implementing focused digital strategies will enhance work efficiency.
Analyze data to enhance product understanding. For smooth integration, standardized data formats are important. To avoid inconsistencies and submission errors, the use of a structured data format by companies will help in streamlining the process.
Test the integration thoroughly before implementing it properly. This includes:
Ensure that data is correctly transferred between eCTD and LMS.
Thoroughly check security measures to protect sensitive information.
Before full-scale implementation, check for small scale submissions to identify errors
Monitor critical indicators, including
Decreased submission errors.
Time saved in regulatory submissions.
By continuously optimizing the system, pharmaceutical companies can stay ahead of regulatory changes and enhance operational efficiency (Macdonald et al., 2021).
As the pharmaceutical industry is changing day by day, integrating eCTD with a labeling management system (LMS) is an absolute need of the time. Due to the increasing demand for faster approvals, pharma leaders can no longer rely on traditional and tedious operations. The integration of structured content management, regulatory submissions, and labeling improves productivity, reduces errors, and shortens time-to-market for drugs. The future holds even deeper integration with emerging technology and increased automation. SCM systems improve regulatory submission management and modernization by enabling machine learning, data mining, and real-time monitoring of patient safety and efficacy.
(Algorri, Cauchon, & Abernathy, 2020; Beierle et al., 2023; Macdonald, Isom, Evans, & Page, 2021; Matsui et al., 2023; Patil & Jogalekar, 2023)
Algorri, M., Cauchon, N. S., & Abernathy, M. J. (2020). Transitioning chemistry, manufacturing, and controls content with a structured data management solution: streamlining regulatory submissions. Journal of Pharmaceutical Sciences, 109(4), 1427-1438.
Beierle, J., Algorri, M., Cortés, M., Cauchon, N. S., Lennard, A., Kirwan, J. P., . . . Abernathy, M. J. (2023). Structured content and data management-enhancing acceleration in drug development through efficiency in data exchange. AAPS Open, 9(1), 11. doi:10.1186/s41120-023-00077-6
Macdonald, J. C., Isom, D. C., Evans, D. D., & Page, K. J. (2021). Digital innovation in medicinal product regulatory submission, review, and approvals to create a dynamic regulatory ecosystem—are we ready for a revolution? Frontiers in Medicine, 8, 660808.
Matsui, R., Yamaguchi, K., Lee, J. J. V., Ting, I., Khairilisani, D., Chang, J., . . . Mo, R. (2023). Survey Result for E-labeling Initiatives in Asia. Ther Innov Regul Sci, 57(2), 251-260. doi:10.1007/s43441-022-00462-5
Patil, N., & Jogalekar, T. (2023). Regulatory affairs: the gateway between the industry and regulatory bodies. Pharm Res(II), 106-114.