
Regulated industries, including pharmaceuticals, medical devices, veterinary medicine, and cosmetics, are operated by regulatory affairs professionals. The regulatory affairs profession focuses on gathering, analyzing, and communicating risk assessments and benefits of healthcare goods to regulatory bodies and the public globally (Rupnawar & Wani, 2024). Pharmaceutical companies are increasingly submitting digital applications instead of paper-based methods, as accepted by industry and health authorities. Currently, the eCTD format is the only worldwide authorized and ICH-recommended standard. Although it has been in use for over 15 years, it is the mandatory filing format for drug applications in Europe and the US and has been adopted by many other health agencies. The Covid-19 epidemic has emphasized the importance of accelerating the global transition to digital formats for regulatory compliance and commercial continuity. It is being adopted by businesses like medical devices, veterinary care, and cosmetics to accelerate submission processes and align with worldwide regulations. Health authorities are engaging with trade associations, enterprises, vendors, and regulators to learn how to apply eCTDs (EFPIA, 2021).
The eCTD is the electronic expression of the CTD as established by the ICH. It includes international guidelines for organizing, formatting, and sending dossiers to health authorities, who can then review them electronically. Industries other than pharma are discovering the positive impacts of eCTD.
● Medical products, including pharmaceutical medications and medical devices, are reviewed and approved by the FDA.
● The FDA regulates a variety of products, including food, cosmetics, and veterinary medications.
● With 20 years of expertise, the European Medicines Agency (EMA) has worked to safeguard the effectiveness and safety of medications for both humans and animals in Europe while promoting innovation and research in their creation. (Lakshmi et al., n.d.).
In today's fast-paced market, a product's and company's success depends on reducing the time it takes to reach a consumer. As a result, the firm's Regulatory Affairs activities must be effectively managed. Improper data reporting can prevent a prompt and successful review of a marketing application. For example, a new drug may have a cost of millions of dollars, and even a delay of a month in a market release might be costly. Incorporating eCTD in sectors other than pharmaceuticals is beneficial, such as (Sanskruti Wagh*1, 2024).
● Improved submission storage and management.
● Improved data arrangement.
● Support for life cycle management.
● Harmonization with global regulatory requirements.
● Access to current and comprehensive information.
● Improved tracking and search tools for reviewers.
● Improved evaluation procedures and transparency.
● Minimal effort and information are required for assessment reports.
● Managing communication with other professionals.
● Optimal use of available resources.
● Improve communication with the industry.
● Optimized business processes.
Implementing eCTD can be complex for the industries that do not have sufficient systems, expertise, and knowledge in this regard.
Although the standardized structure of eCTD has many benefits, there are certain drawbacks that make it challenging for different organizations. Finding regulatory tools to help software compliance with eCTD submission requirements is a serious challenge.
To properly submit an eCTD, companies must have all documents, including types of products, responses, revisions, and restorations, in a digital form (Ahammad et al., 2019).
Current electronic publishing software extracts links, bookmarks, and navigational headings from source files (such as Word, PDF, etc.) to speed up publishing. However, if the files are not properly formatted, manual modifications may be necessary. Companies that handle submissions in-house employ stringent rules and templates to quicken the process. If any of these functions are carried out externally, obstacles increase, and it is worth considering the following suggestions:
● Establish standard procedures, templates, and forms and share them with external service providers.
● Publishing departments/providers should define and communicate source file specifications to all parties involved.
● Test your source files in publishing software prior to final publication.
To avoid uncertainty in the publishing process, regulatory affairs and publishing staff must communicate clearly. The eCTD submissions rely on metadata, which must be entered accurately to avoid delays or errors. Ambiguous or missing information may hinder development or lead to misunderstandings, necessitating time-consuming revision. The use of defined procedures and forms guarantees that publishing runs smoothly.
Medical devices, veterinary products, and cosmetics deal with varied regulatory requirements, unlike pharmaceuticals, which have well-defined global eCTD standards. Some agencies require eCTD, while others accept hybrid or paper-based submissions, resulting in discrepancy.
Software and IT infrastructure contracts must be executed on both the health authority and industry sides. To reduce adoption errors, material to facilitate the transition must be easily available on the health authority website, and open contact between the health authority and industry must be ensured at regular time intervals. Set defined times and prior notification of changes to ensure compliance (EFPIA, 2021).
Digital disruption presents a challenge for firms to upskill their workforce. Effective digital change management requires a culture of constant learning, an agile mindset, and including all workers in the transformation journey. To successfully transition, companies must assess their leadership styles, personnel competencies, and incentives for ongoing professional growth (Chisholm & Critchley, 2023).
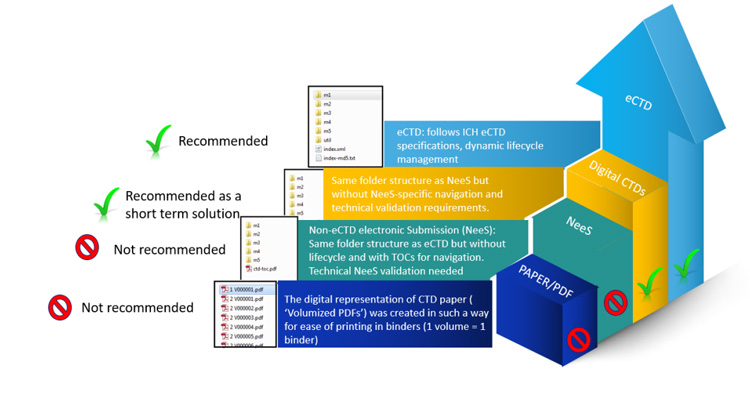
Figure 2. Stages involved in eCTD adoption (EFPIA, 2021)
Different sectors of life sciences encounter the complexities of eCTD implementation. Willingjet Life Science plays a very essential role in overcoming the challenges associated with this transition of regulatory submission.
Achieving multilingual compliance expertise in medical, technical, and regulatory translations ensures that labels, packaging inserts, and documentation comply with region-specific standards.
From document creation to regulatory submission tracking, Willingjet guarantees that eCTD dossiers meet all compliance criteria, reducing the possibility of rejection or delay.
Due to insufficient infrastructure and various regulatory requirements, many industries in Southeast Asia, the Middle East, and Latin America encounter difficulties in implementing eCTD. Willingjet provides tailored solutions to handle difficulties and comply with regulatory requirements.
Willingjet offers consulting eTCD services to enterprises that are inexperienced with eCTD, assisting them in understanding eCTD regulations, adopting best practices, and simplifying regulatory workflow.
The eCTD is becoming a standard procedure in various businesses with changing regulations. eCTD is a significant future advancement in submitting information for medical devices, cosmetics, and veterinary medicines.
● With the help of eCTD exchange of regulatory information between industries and agencies, the development, review, and management of electronic submissions take place.
● Regulatory organizations align eCTD requirements, resulting in improved harmonization across the globe.
● Companies may soon be able to make submissions to multiple regulatory authorities with a single keystroke (Mahaparale & Desai, 2018).
Many other industries, like the pharmaceutical industry, depend on regulatory affairs to ensure product safety, efficacy, and quality before reaching the market. Regulatory officers serve as the main interaction between companies and regulatory bodies, ensuring compliance with evolving rules and regulations. Regulatory work ensures public health and facilitates the global approval and commercialization of a wide range of products, including medical equipment, veterinary medicines, and cosmetics. As medication research and market access become more complex, eCTD, a coordinated regulatory strategy, is crucial.
With our Willingjet Life Sciences team of professionals, we ensure high-quality translation in a variety of medical sectors. If you need experienced medical translations for your regulatory documents, contact us right away to accelerate your submission procedure.
Ahammad, N., Reddy, N., Nagabhushanam, M. V., & Ramakrishna, B. (2019). Challenges faced during ectd and ctd filling procedures for usfda and canada. Journal of Drug Delivery and Therapeutics, 9(4-s), 673–679. https://doi.org/10.22270/jddt.v9i4-s.3334
Chisholm, O., & Critchley, H. (2023). Future directions in regulatory affairs. Frontiers in Medicine, 9, 1082384. https://doi.org/10.3389/fmed.2022.1082384
EFPIA. (2021, December 20). Global ICH eCTD Adoption Efpia https://www.efpia.eu › media › ectd-white-paper.
Lakshmi*, D. A., Nagabhushanam, M. V., & Ramakrishna, G. (n.d.). DOSSIER PREPARATION REQUIREMENTS FOR GENERIC DRUGS OF USA, EUROPE & INDIA. World Journal of Pharmaceutical Research. Retrieved March 24, 2025, from https://www.wjpr.net/abstract_show/23559
Mahaparale, S., & Desai, B. (2018). Role and overview of drug regulatory affairs in pharmaceutical industry with implementation of CTD and eCTD. World Journal of Pharmaceutical Research, 7(7), 201–215. https://wjpr.s3.ap-south-1.amazonaws.com/article_issue/1522475057.pdf
Rupnawar, M., & Wani, P. (2024). Pharmaceutical Regulatory Affairs: An Overview of Global Regulatory Frameworks and Emerging Trends. Int. J. Sci. R. Tech, 1(12). Sanskruti Wagh*1, W. S. U. 2. (2024). Understanding regulatory affairs in the pharmaceutical industry: Roles, importance, and global perspectives. https://doi.org/10.5281/ZENODO.12205641